Editor's Note: This article has been updated to reflect the correct amount of recommended rates of Boron (B), .875-1.0 ppm. We apologize for the error.
Plants require certain micro and macronutrients to thrive. Although plants do not need micronutrients in the same concentration as the meat-and-potatoes macronutrients, they are just as important for plant growth, development and biomass production. Micronutrients iron (Fe), boron (B), manganese (Mn), zinc (Zn), copper (Cu), and molybdenum (Mo) are all essential to produce healthy plants. With the recommended rates of micronutrients being smaller in comparison to macronutrients (nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S)), it is easy for growers to overapply them. But just because plants require these essential elements in lesser amounts doesn’t mean an imbalance won’t have an outsize impact on crop health and yield. Excess application of micronutrients can cause nutrient antagonism and imbalance, which limits nutrient concentration and uptake, meaning key elements are unavailable to plants. There is a specific range where micronutrient fertility is optimized, but not overapplied.
In plants, most all micronutrients are immobile elements, meaning they cannot be translocated from sufficient to deficient plant parts. (The exception is Mo, which is partially mobile.) Plants use micronutrients for various processes, including B for cell wall formation and Fe for chlorophyll formation and cell division. Given the diverse roles micronutrients play in plant development, these directly affect optimal growth and yield of cannabis. Because cannabis is a determinate plant, meaning once flowering begins, the terminal growing tips develop into floral material, growers should closely monitor anything that would impact plant growth during production to maximize floral development and yield.

Causes of Micronutrient Toxicity
Micronutrient toxicities can be caused by multiple factors, including a fertilizer regime with excess concentrations of these elements. This can happen because of an initial mixing error where a stock tank’s concentration is too high, or an improperly calibrated injector that overfeeds the plants.
Toxicity Prevention
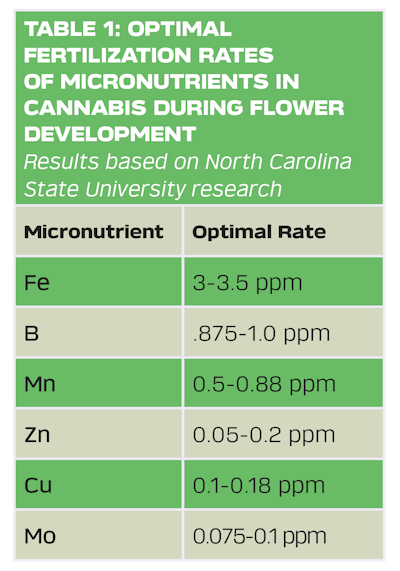
However, there are many steps growers can take to prevent toxicity and balance fertility, such as understanding the narrow ranges of micronutrient fertility (Table 1), regularly sampling irrigation water, and monitoring substrate/fertilizer solution pH weekly. Substrate pH is the root of various problems for cannabis growers. When substrate pH falls below 5, increased micronutrient availability can occur, potentially leading to excessive, toxic uptake of Fe and Mn. Monitoring substrate pH to keep it within the recommended range of 5.5 or higher helps to ensure optimal root zone conditions are maintained to maximize plant growth. Adding liming materials to soil or substrate is one of the most common methods to raise substrate pH, as it reduces the toxic effects of exchangeable aluminum (Al) and Mn ions and increases the base saturation of the substrate (For more on correcting pH, read: “New Research Results: Optimal pH for Cannabis").

What Does Micronutrient Toxicity Look Like?
During research trials at North Carolina State University (NCSU), the authors documented micronutrient toxicity symptoms and leaf tissue nutrient ranges to help growers identify the disorders.
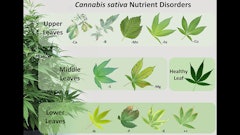
Boron Toxicity In trials, marginal chlorosis (yellowing) of the lower foliage (see Fig. 1, above) was the first boron toxicity symptom. The yellowing then intensified and spread inward toward the leaflet midrib. In severe cases, leaf margins became brown and eventually necrotic (dead). The NCSU study reported that B toxicity symptoms first appeared when the foliar leaf concentration of B was 671.75 ppm after plants received a fertility 10 times the recommended B rate of .875 to 1.0 ppm. Plants that received the recommended fertility rate had foliar leaf concentrations of 64.6 ppm B when grown in a neutral pH substrate.
Manganese Toxicity Manganese toxicity initially developed as marginal tip burn on the lower foliage and spread to the leaf midrib as symptoms advanced (Fig. 2). In severe cases, leaves exhibited necrosis and dropped (abscised) from the plant when ranges exceeded the optimal fertility rate of 0.88 ppm Mn.
Iron Toxicity While Fe toxicity can be caused by overapplication, it most often occurs when the pH falls below 5. Plants with Fe toxicity initially had discolored, dark brown leaves, known as “bronzing” on the lower foliage (Fig. 3). As micronutrient toxicities develop, they will begin in the lower, older foliage and spread into the upper foliage. In severe cases, leaves became necrotic and abscised. The recommended fertility range for Fe is 3 to 3.5 ppm.
Optimal Micronutrient Fertility Rates
Given the study results, NCSU researchers believe the optimal fertilization rates of micronutrients for cannabis during flower development are: 3-3.5 ppm Fe, .875-1.0 ppm B, 0.5-0.88 ppm Mn, 0.05–0.2 ppm Zn, 0.1–0.18 ppm Cu, and 0.075-0.1 ppm Mo (see Table 1, above) To help avoid accumulation in the plant, growers should couple these feeding rates while monitoring the substrate or fertilizer solution pH weekly to ensure that it is above 5.5.
Corrective Measures
Excessive nutrient accumulation in the leaves cannot be mitigated. If appropriate for your production system, use clear water to leach out the excess nutrients to lower the levels. Applying Ca, specifically calcium nitrate, can help tie up some of the excessive micronutrients. This application will help increase the substrate pH, tie-up micronutrients, and provide required Ca and N to the plant. Growers should also monitor all parts of their production system regularly, outlining steps in standard operating procedures (SOPs). Monitoring substrate pH also is key to prevent it from becoming too low. If the substrate pH falls below 5.5, growers can utilize flowable lime to raise levels.
When micronutrient toxicities are suspected, play it safe and submit a leaf tissue sample for analysis to confirm the toxicity before radically altering your fertility regime.