For controlled-environment-grown crops, substrate pH and electrical conductivity (EC) are two main factors that can easily be monitored to make sure your crop is on track for success. This monitoring principle has been used by the commercial floriculture industry for the past 40 years, and it can be adopted by the cannabis (Cannabis sativa) industry, too. Monitoring and managing the substrate pH and EC could help to avoid up to 80 percent of the plant nutrition problems that occur with controlled environment crops. (For more on optimizing pH, see the first article in North Carolina State University's (NCSU) ‘Nutrient Matters’ series in the March 2019 issue of Cannabis Business Times.) In this column, we’ll discuss what EC is, why it’s so significant to crop health and how to measure it.
What is EC?
Electrical conductivity is the electrical charge that moves through a solution. The higher the salt concentration, the greater the electrical reading. This principle is applied to controlled environment production because the higher the concentration of fertilizer salts in the solution or in the substrate, the higher the EC reading will be. We can then monitor the EC level to ensure that fertility levels are where they need to be.
What EC Does (and Doesn’t) Measure
The EC is a measurement of the total amount of fertilizer salts in the solution. It does not provide specific information about each individual element or that individual element’s concentration in solution.
The good news is that with controlled environment production, the primary nutrients we provide are nitrogen (N), phosphorus (P) and potassium (K). Therefore, with most cases in both theory and practice, the EC reflects the relative concentration of those elements. You can apply this concept to cannabis to confirm the accuracy of the fertilizer concentration you apply in the solution and to monitor the crop’s nutritional status.
Lower-than-optimal EC readings typically indicate a nutrient deficiency, especially low levels of N (Fig. 1). High EC levels can indicate you are supplying too much fertilizer or that your plants are not absorbing the nutrients, both of which can lead to a salt toxicity leaf burn.
Measuring the Fertilizer EC
Doing the Math
If you are using a fertilizer such as 13-2-13 Cal-Mag, the label on the back of the bag provides EC guidelines to help you double-check the accuracy.
For example, if you want to provide 200 ppm of N to the crop from a 13-2-13 Cal-Mag solution, then you would need to mix 20.76 ounces in 100 gallons of water. (This is based on Jack’s brand fertilizer. Manufacturers use different salts to achieve a similar analysis, so you need to utilize recommendations on their labels to be correct.) Alternatively, if using an injector set at a 1:100 ratio, that would mean adding 20.76 ounces of Jack’s Cal-Mag into a gallon of water to make a concentrate that is then run through the injector. Either way, the corresponding EC of that final diluted fertilizer solution should be at 1.64 millisiemens per centimeter (mS/cm, the unit by which EC is measured), as listed on the product’s label.
By measuring the EC, you can double-check your mixing procedure and confirm if your fertilizer injector is working accurately.
Accounting for Irrigation Water EC
You must also take into account the EC contribution of your irrigation water. If your irrigation water has high concentrations of alkalinity (calcium (Ca), magnesium (Mg) or sodium (Na)) then when you test the EC of your fertilizer solution or substrate, your values will be artificially elevated, too. When monitoring the fertilizer solution you are supplying to the plants, knowing the EC contribution of your irrigation water is important. This requires that you periodically have your water quality tested by a commercial lab.
In Raleigh, N.C., we have very pure water, and normally the EC is around 0.11 mS/cm. Therefore, we must add the EC contribution of the irrigation water to the EC contribution of the fertilizer to determine the corrected target EC. This would mean the final fertilizer solution EC should be 1.75 mS/cm (1.64mS/cm fertilizer contribution + 0.11 mS/cm water contribution). In contrast, for much of the Midwest, the EC of the irrigation water is much higher. If the water EC was 1.13 mS/cm, the same final fertilizer solution EC would instead be 2.77 mS/cm (1.64 mS/cm fertilizer contribution + 1.13 mS/cm water contribution) to also provide 200 ppm N from a 13-2-13 Cal-Mag fertilizer by Jack’s.

Accuracy Check
Your fertilizer solution EC value can be used to check for fertilizer delivery accuracy. If it is not within 5 percent, then you need to double-check the mixing math, the accuracy of measuring out the fertilizer salts, or if the injector is malfunctioning. The EC values will be significantly lower as the injector begins to fail, which in turn will slow growth for plants grown in soilless substrates and, under prolonged conditions, cause lower-leaf yellowing due to inadequate fertility.

Hydroponic/Rockwool System
If you are growing a cannabis crop hydroponically or in an inert substrate such as rockwool, you can monitor the fertilizer EC value to ensure you are providing the correct fertilizer concentration.
Measuring the Substrate EC
If you are utilizing a soilless substrate containing sphagnum peat moss or pine bark, you can use another aspect of EC monitoring to make sure your fertilization program is on track.
Begin an In-House Nutrient Monitoring Program
A wide assortment of pH and EC meters is available on the market. At NCSU, we recommend combination pH and EC meters. The majority of growers we work with purchase a unit that costs less than $200.
Monitoring and managing the substrate pH and EC could help to avoid up to 80 percent of the plant nutrition problems that occur with controlled environment crops.
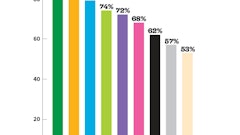
After purchasing a combination pH and EC meter, you need to start using it! The majority of the commercial floriculture greenhouse industry relies upon the non-destructive PourThru monitoring method to test EC and pH. Weekly or biweekly sampling is the key to ensuring your EC and pH levels are on track. While cannabis-specific recommendations based on scientific research have not been published, sampling procedures (including the PourThru method) can be found at the nutrient monitoring website: fertdirtandsquirt.com and general EC guidelines are provided in Fig. 2 above.
Target Fertilization Rates and EC Levels for Stages of Plant Development
The nutrient demands of plants change over the course of development. Young transplants focus energy on establishing roots and add little leaf growth; as such, they require less fertilizer. As plants grow and bulk up, fertilizer needs increase. The number of leaf and flower nodes on each plant sets when the photoperiod changes and flower formation begins. This terminal inflorescence (flower production) establishes how much more growth will occur. At this stage, the amount of vegetative mass the plant adds is less, yet energy and resources are still required for flower/bud development. With this shift from vegetative growth to flower growth, the nutrient demand lessens. Given how a plant grows and adds mass, and how nutrient demands vary over time, it comes as no surprise that nutrient delivery needs to be customized to meet the changing demands of the crop throughout its different growth stages.
Your fertilizer solution EC value can be used to check for fertilizer delivery accuracy.
The EC is an indirect measure of the fertilizer salts contained in the substrate, so those target levels will also vary at different growth stages. By starting an in-house nutrient monitoring program and measuring the EC, you can make sure that sufficient levels of fertilizer are available to the plants. Recommended fertility levels and target EC levels based on the stage of cannabis plant development are listed in Fig. 2, as measured by the PourThru method.
Ammoniacal-Nitrogen, Urea and Organic Fertilizer Adjustments
Though measuring the EC is an excellent tool when utilizing water-soluble controlled environment fertilizers, there are limitations to this measurement. When it comes to measuring the contribution of N to the EC, nitrate-nitrogen (NO3-N) can be measured; however, the other two common forms of nitrogen included in fertilizers—ammoniacal-nitrogen (NH4-N) and urea—cannot be measured by EC. The EC charts on the fertilizer bag, or provided by the manufacturer, will reflect those lower target EC values. Likewise, most organic fertilizers rely upon ammoniacal-nitrogen (NH4-N) and urea for their derived sources of N. Be aware of this fact when you are mixing the fertilizer solution. In addition, you must then adjust down the target EC ranges accordingly when testing the substrate EC.

The Bottom Line
Optimizing fertility management is a strategy of supplying the appropriate level of nutrients to cannabis to meet the demands for plant growth. If the supply is too low, then the genetic potential of the plant is hindered because of the deficiency, and the plant will flower less. Elevated fertilizer levels can lead to excessive leaf and shoot development at the expense of flower growth. Excessively high levels of fertilizer in the substrate can also lead to stunted growth.
The good news is that conducting periodic PourThru sampling of both the EC and the pH will help you monitor the nutrient status of your crop to ensure it is on track.
Brian Whipker, Turner Smith and Paul Cockson are from Department of Horticultural Science, North Carolina State University, Raleigh, N.C. Hunter Landis is from North Carolina Dept. of Agriculture and Consumer Services, Raleigh, N.C.