
Reviewed December 2023
The recommended substrate pH for cannabis (Cannabis sativa) varies widely. A recommended substrate pH of 6.0 to 6.5 often is cited as the standard. However, we have observed pH ranges between 4.0 and 5.0 or greater than 7.0 during our visits to commercial greenhouses growing cannabis.
Our observations from those greenhouses indicated that plants grown at acidic (lower) pH appear to have normal growth and lack the symptomology of lower leaf blackening or bronzing typically seen with other greenhouse species at lower pH levels. In crops grown at higher pH, only in rare instances did we observe interveinal chlorosis of the upper (youngest) leaves (a common symptom of greenhouse crops grown at high pH). These observations suggest that cannabis is not prone to iron deficiencies when the substrate pH nears 7.0.
Returning to our labs at North Carolina State University (NCSU), we began our work to understand the underlying factors affecting pH in substrates and how those fluctuations impact the Cannabis sativa plant in order to determine the optimal pH range for cannabis.
This is what we discovered.
pH on the Range
Substrate pH is important to plant nutrition because it directly impacts plant nutrient availability.
Substrate pH levels below 5.0 result in increased micronutrient availability that can lead to iron (Fe) toxicity or manganese (Mn) toxicity, or both. Symptoms of Fe toxicity and Mn toxicity may appear as black speckling on older leaves (observed with zinnias or gerbera) or as bronzing (observed with marigolds or seed geraniums). While some crops display low pH symptoms on lower leaves, others may only display low pH through stunted growth, as is the case with poinsettias.
In our initial experiments at NCSU, we did not observe symptomology of lower leaf blackening or bronzing on Cannabis sativa plants, only stunted growth. These preliminary results suggested that cannabis can regulate micronutrient uptake under low substrate pH conditions similar to a poinsettia.
When the substrate pH becomes too high, many species develop interveinal chlorosis (yellowing) on the youngest leaves. This is a common situation that occurs with many greenhouse-grown species (such as petunias) when the elevated pH makes micronutrients, such as Fe, unavailable to the plant, even if adequate levels are provided in the fertilizer.
We did not observe symptomology of interveinal chlorosis in cannabis plants grown at pH 7.0 in our initial experiments at NCSU, either. Iron uptake can also be hindered by over-irrigation, cold substrate temperatures or root disease such as Pythium. In other experiments at NCSU, we did observe the development of interveinal chlorosis on plants with a pH of 7.8. This observation provides more refinement of the upper pH limit and would suggest that the pH should be lower than 7.5.
The preliminary results of NCSU’s research imply that vegetative stock plants of cannabis have a wide substrate pH range in which the plants will optimally grow. That range appears to be as wide as pH 5.0 to 7.0. Based on experience with other species, a narrower range of 5.5 to 6.5 may be more appropriate to target, as this will allow growers to more easily adjust as pH approaches the ends of the targeted range.
For growers, utilizing these pH values in a monitoring system such as the PourThru method implies that the safe pH zone to target would be a narrower 5.8 to 6.2. If pH drops below 5.8 or above 6.2, corrective procedures should begin to adjust the pH back into the 5.8 to 6.2 range.
(Note: These values are for plants grown in greenhouse conditions with a soilless substrate (growing medium). Optimal ranges for field-based growing conditions typically would move up the lower and the upper range by 0.5 pH units to 6.2 to 6.7—as field soils latch on to more nutrients than a soilless substrate such as peat or coir, and risk of nutrient lock-up is lessened.)
 newsletter subscribe
newsletter subscribe
How to Correct pH
Again, when the pH drifts into unwanted territory, adjustments must be made. Below are standard corrective procedures used to modify substrate pH for plants grown in soilless substrates in greenhouses and adapted for cannabis.
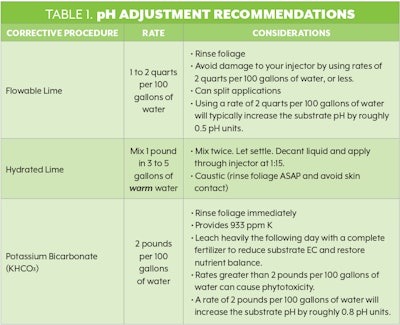
1. Low Substrate pH Correction When Fe toxicity and Mn toxicity become a problem, raise substrate pH to the recommended pH range. Corrective procedures to raise low pH levels are listed on p. 52 (Table 1). Switching to an alkaline fertilizer when substrate pH is nearing the lower limit will help stabilize the pH.
Flowable Lime
To make small adjustments of roughly 0.5 pH units, mix 1 to 2 quarts of flowable lime per 100 gallons of water. If using an injector, avoid using higher concentrations of flowable lime as it will damage it. If higher rates are needed, then split your applications to avoid damaging your injector. To avoid leaf burn, it is best to rinse the foliage after treatment if any flowable lime comes in contact with the leaves.
Hydrated Lime
For more stubborn low-pH problems, use a hydrated lime mixture. Mix 1 pound of hydrated lime in 3 to 5 gallons of warm water. Mix it twice and let it settle after each mixing. Once the sediment collects in the bottom of the container, pour the liquid through your injector system set at a 1:15 ratio. This product is corrosive, so rinse the foliage as soon as possible to avoid leaf burn, and avoid skin contact.
Potassium Bicarbonate (KHCO3)
Potassium bicarbonate should be handled with care. It can throw off your substrate chemistry as it provides 993 ppm of potassium (K) in mixtures of 2 pounds per 100 gallons of water. Rinse the foliage immediately after application via injectors and leach heavily the following day with a complete fertilizer to reduce substrate electrical conductivity (EC) and restore nutrient balance. The 2-pounds rate will increase the substrate pH by roughly 0.8 pH units. Rates greater than 2 pounds per 100 gallons of water can cause phytotoxicity.
As always, remember to recheck your substrate pH to determine if reapplications are needed.
2. High Substrate pH Correction The target pH for cannabis is between 5.8 and 6.2. Higher pH values may result in Fe deficiency and create interveinal chlorosis on upper leaves. Check the substrate pH to determine if it is too high. Be careful when lowering the substrate pH, as low pH can be more problematic and difficult to manage.

Acid-Based Fertilizer
If the substrate pH is beginning to increase, consider switching to an acidic-based fertilizer. These ammoniacal nitrogen (N)-based fertilizers are naturally acidic, and plant nitrogen uptake will help moderate the substrate pH over a week or two.
Acid Water Drench
Some growers use this intermediate correction when pH levels are not excessively high and a quick lowering of substrate pH is desired. Sulfuric acid is recommended to acidify irrigation water to a pH 4.0 to 4.5. Apply this acid water as a substrate drench, providing 5 percent to 10 percent excessive leaching of the substrate. Rinse the foliage to avoid phytotoxicity. Results should be visible within five days. Retest the substrate pH and repeat if needed.
Iron Drench
If the levels are excessively high, then a Fe chelate application can be made to the substrate.
- Iron-EDDHA can be mixed at a ratio of 5 ounces per 100 gallons of water.
- Iron-DTPA can be mixed at a ratio of 5 ounces per 100 gallons of water.
- Iron sulfate can be mixed at a ratio of 4 ounces to 8 ounces per 100 gallons of water.
Apply the iron as a substrate drench with enough volume to leach the pot, and rinse the foliage immediately after application.
Conclusions
Based on grower observations and initial experiments at NCSU, it is possible to grow cannabis with a wider soilless substrate pH range than most other species. Cannabis plants do not appear prone to develop leaf symptomology when substrate pH is too low or too high compared to the current general greenhouse standards—only plant stunting occurs at sub-optimal conditions.
Therefore, based on research and experience with other species, a wider range of 5.5 to 6.5 may be used. When adapting these values to a monitoring system, the recommended pH zone to target would be 5.8 to 6.2. By monitoring the substrate pH over time, one can assure that plants are within the optimal range.
Brian Whipker, Turner Smith and Paul Cockson are from Department of Horticultural Science, North Carolina State University, Raleigh, N.C. Hunter Landis is from North Carolina Dept. of Agriculture and Consumer Services, Raleigh, N.C.



















