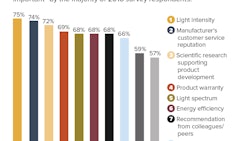
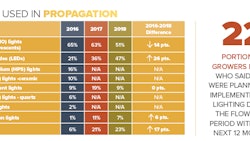
Given the shortage of proven cultivation knowledge and data, the big opportunity lies in learning faster than the competition. One critical skill for accomplishing that is knowing how to design an experiment. This article explains that process using a lighting research project as a case study. The project was based on a research partnership between Greenseal Cannabis Company, Voltserver Inc. and Université Laval. Several lighting companies also contributed, including BIOS Lighting, Fluence Bioengineering, Illumitex and P.L. Light Systems.
Experimental Design
A controlled experiment is the process of manipulating conditions and collecting data to answer a question. For example, will increasing CO2 from 1,000 ppm to 1,100 ppm increase yields? The “design of experiments” (DOE) is the process of planning the experiment to ensure that you are generating the right data (and enough of it) to answer your question.
Developing a Hypothesis
The DOE process starts with a question to which the answer is important to your business. Each experiment we perform is the output of a structured process for identifying and ranking potential research opportunities. We identified lighting as a top opportunity because it is:
- one of the grower’s largest capital expenses,
- likely the single most important factor for yields and quality, and
- there’s been almost no rigorous academic cannabis lighting research.
Literature Review
We started by reviewing the research (academic and industry) on the topic. For instance, spectrum research for other plants indicates that a grower could harm the plant’s development by excluding certain wavelengths, but otherwise suggests that if a light provides a broad spectrum (like a multivitamin), then the plant will take what it needs. But we wanted to know: How do spectrum quality differences across full-spectrum lights impact yields?
Regarding the impact of intensity, researchers documented in a 2008 study published in the journal Physiology and Molecular Biology of Plants that cannabis net photosynthesis increased with light intensity up to a PPFD (photosynthetic photon flux density, a measure of light intensity) of as much as 2,000. This is about five times more intense than a double-ended HPS mounted 36 inches above the canopy. This suggested the possibility of substantially increasing yields by substantially increasing light intensity.
Yet the researchers did not answer our question because research has also shown that measuring changes in net photosynthesis is not a reliable way to estimate changes in final yields. One industry study by Fluence Bioengineering, however, did find substantial yield increases resulting from higher PPFD. So, our second question we wanted to address was: What is the relationship between high light intensity and yields?

Research Methods
A well-defined research question makes it easy to define the data you need to answer the question confidently (in a statistical sense). For instance, imagine you increase CO2, and your yields increase by 5 grams per square foot. How confident are you that increasing CO2 increased yields? If yields typically vary by roughly 4 grams from one harvest to the next, then maybe you wouldn’t feel confident. If the typical variation was 1 gram, then you should be more confident. Then if you increased CO2, but also happened to change the process for taking clones, you might wonder how much of the yield increase was caused by the CO2 and how much by the new cloning method. My point is that if your question is well defined, then there is a standard protocol you can follow to help you feel confident about the final answer implied by your experimental results.
We wanted to measure the impact of light intensity and spectrum on yields. We decided to manipulate these factors by using several different types of commercially available lights that vary in intensity and spectrum. These included double-ended HPS, broad-spectrum LEDs and purple LEDs. Eight different models in all provided varying spectrums and intensities at the canopy that ranged from around 450 PPFD to an average PPFD of nearly 2,000. Light intensities were measured using a quantum sensor.
Control and Treatment Groups
Next, we needed to grow individual tables of plants under the different lights. Our reference group, or control group, would be plants grown 36 inches under double-ended HPS lights. The other table-light combinations are called treatments. The environmental conditions for each treatment and the control are kept as similar as possible, except for the lights.
Randomization
To account for the fact that clone health and potential will vary, we randomly assigned clones to each table. This is called randomization.
Number of Replicates
This brings us to one of the most critical steps in the DOE process: How many times should we grow groups of plants under each light? In other words, should we have one table growing under Light A or more? This is the number of replicates or repetitions of the experiment, and to be sufficiently confident about your final results, you need a sufficiently high number. Otherwise it’s impossible to rule out that some other confounding factor (e.g., Treatment 1 was randomly stocked with healthier clones) drove your results. (Note: This is one way to calculate the number of repetitions to conduct. An experienced consultant may be able to help configure the number of times most suitable to a cultivator’s particular environment.)
To select the number of replications, researchers commonly use the following formula:
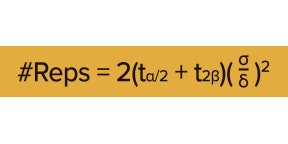
Where: ta/2 and t2ß are “t values” associated with the probability of a Type I error (a in the formula) and a Type II error (ß in the formula). A Type I error (a “false positive”) occurs when we detect differences that don’t actually exist, whereas a Type II error occurs when we fail to detect a difference that does exist. d is the minimum difference you’d like to be able to detect confidently, and s is the standard error in differences (e.g., typical variation in yields) observed across similar past experiments.

Example
The probability of a Type I error (a) is typically set at 5%, while 10% or 20% is typically used for a Type 2 error (ß). No theory is behind this—it’s just convention. Suppose we used 5% and 20%, respectively. From growing the same cannabis strain under similar conditions in the past, we estimated the standard error in yields to be 30 grams. If you do not have past data to use, you have two other options: You can use data from other researchers for comparable experiments, or you can do a pilot study to create your own estimate. Finally, assume we want to be able to detect a yield difference as small as 60 grams per table.
Step 1: The first step is to find ta/2. An easy way is to use the TINV() function in Excel. The function takes two inputs: a and the “degrees of freedom.” The calculation for the degrees of freedom changes with your experimental design, but if you’re completely randomizing your plants, then you can use the number of plants per treatment as an estimate. So suppose you have 64 plants per treatment, then you would input “=TINV(0.05,64),” which gives you 1.997.
Step 2: Find t2ß by inputting =TINV(2*(1-ß), 64) = TINV(2*(1-0.2), 64), which gives you 0.847.
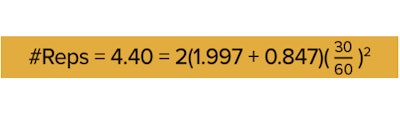
So for this example, we would need five replications per treatment. In other words, for each light-table combination, we needed to repeat the experiment a minimum of five times.
Control
There are many ways to conduct experiments—by changing multiple factors at once and conducting a statistical analysis, or changing a single factor and observing its impact on the plant environment. In our experiment, we chose to manipulate a single factor: light intensity. So to help maintain similar growing environments across the tables, we kept them all in the same room and minimized light from spilling over from one table to another using panda film. We added fans and kept one side of the tables open to increase airflow. We monitored temperature, humidity, CO2 levels and fertigation. Since plants that grow faster will consume more water and nutrients, we’ve recently added tensiometers (which measure soil moisture) and data loggers, so we can be sure that watering occurs when a treatment’s soil moisture reaches a certain threshold. You will also want to employ a CO2 meter to ensure it is being distributed to the crop adequately during the trial.
Results
Our experiment’s results run counter to the current industry paradigm. In our facility, using HPS lights at 36 inches provides around 450 PPFD. The exact PPFD depends on the spillover from other tables. But the highest yields in our experiment resulted from using a full-spectrum LED that provided an average PPFD of nearly 2,000. Specifically, we have been able to increase yields from between 40% and 70% above our control by increasing light intensity up to nearly 2,000 PPFD. This lighting configuration uses more electricity compared to HPS, but for every dollar we spend on more electricity to increase light intensity, we increase the value of our yields by as much as $8 CAD.
Finally, the results of our tests showed that spectrum doesn’t appear to matter much for yields when comparing full-spectrum lights. Thus, at Greenseal, rather than focusing on metrics such as dollars per watt, or searching for a magic spectrum, we simply select full-spectrum lights that minimize the cost of delivering our target PPFD to our plants.